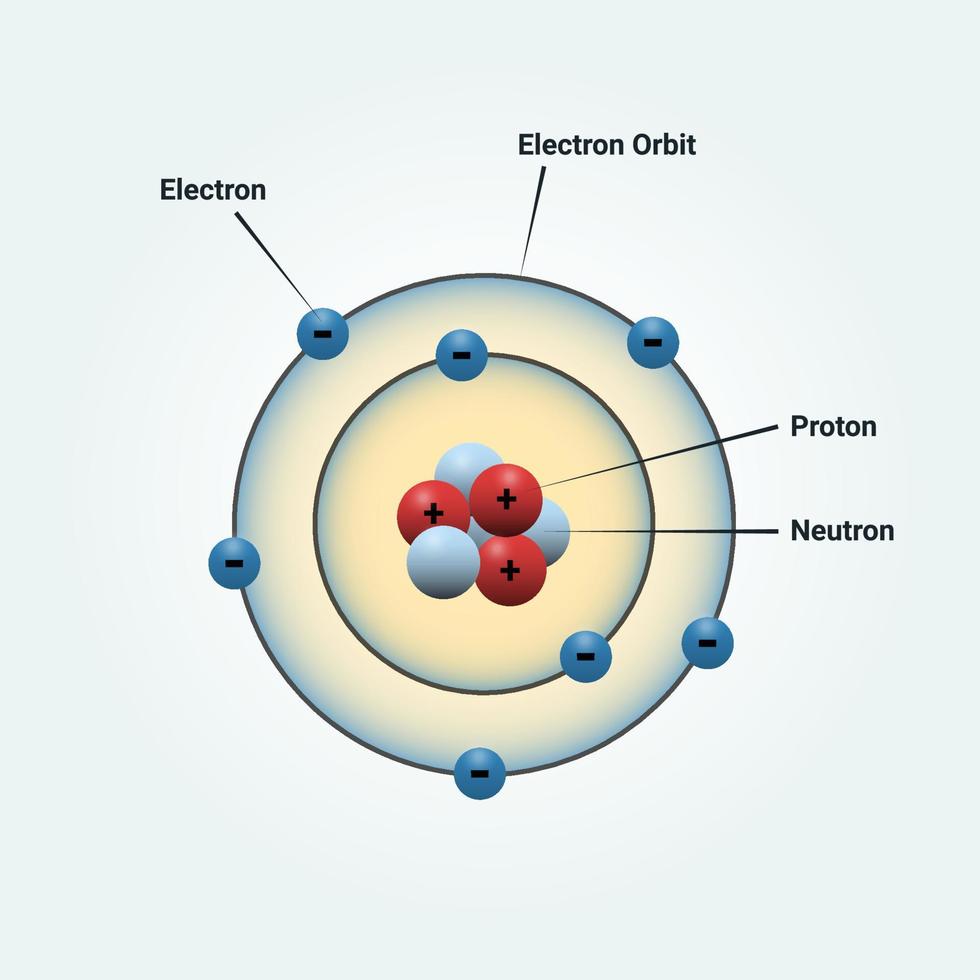
Bohr proposed his quantum mechanical model of the atom in 1913.The energy of an electron was fixed or quantized into discrete values based upon its current orbit. Electrons that were close to the nucleus had lower energies than ones orbiting at a distance. He also proposed that electrons had very specific energies associated with each orbit.Electrons could exist in one orbit or another, but nowhere in-between.  
Bohr proposed that electrons are orbiting, but that they can only orbit at specific predefined distances from the nucleus.'Bohr had made use of a simple and regular pattern for the spectrum of the. 'Bohrs model of 1913 for the hydrogen atom had circular electron orbits about the proton like Earth orbits around the sun,' says Herschbach. For reasons beyond the scope of this course, electrons would emit energy as they rotated around the nucleus, eventually spiraling into the nucleus. Niels Henrik Bohr (1885-1962) was a Danish physicist who developed the atomic model and won the 1922 Nobel prize in physics. However, there were problems with this idea. Among these pioneers, however, one person stands out as particularly important: Niels Bohr. The study of atoms goes back centuries, with scientific pioneers like Dalton, Thomson, and Rutherford each making significant contributions to the field. One of the possible models proposed by Rutheford after completion of his gold foil experiment stated that electrons orbit the nucleus like planets orbit the sun. Spread the loveOne of the most fundamental principles of chemistry is an understanding of atomic structure.Atomic Theory: Niels Bohr Niels Bohr (1885 - 1962)
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
